Managing genetic diversity in relocated Golden Bandicoots

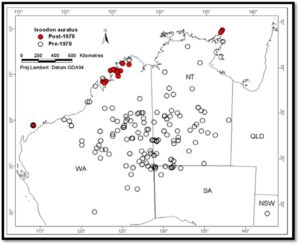
At a recent workshop which investigated genetic considerations for fauna translocations, Dr Kym Ottewell explained that the effectiveness of fauna reintroduction programs is often limited by the availability of source animals. In particular, genetic diversity often declines along with associated fitness in relocated populations that have small source populations and/or maintain small long-term population size due to founder effects, genetic drift, and inbreeding. Dr Ottewell’s presentation utilised the recent translocation of Golden Bandicoots by the Department of Parks and Wildlife (DPaW) to the ex-pastoral lease at Lorna Glen into a predator proof enclosure as her case study example. Golden Bandicoots (Isoodon auratus) were once widespread on the mainland, but are now confined to Barrow Island, Marchinbar Island and the Dampier Peninsula in the Kimberley (Ottewell et al. 2014).
In addition to the recently translocated population at Lorna Glen there are two other translocated populations established at Hermite and Doole Islands off the Western Australian coast.
Figure 1. Past and present geographic distribution of naturally occurring populations of Golden Bandicoots (Modified from Palmer et. al, 2003. Northern Territory DIPE; Ottewell presentation at Genetic Diversity workshop 12/5/16)
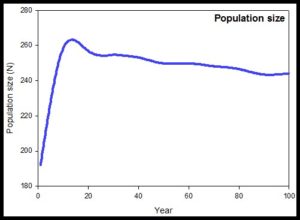
Golden Bandicoots are abundant on Barrow Island, so there was significant genetic diversity in the source population, and they have the capacity to reproduce quickly which should enhance the long-term maintenance of this genetic diversity in the translocated population. Seventy eight male and 82 female Golden Bandicoots were released into the 1,100ha predator proof compound at Lorna Glen in February 2010. The population initially rapidly increased as expected and has now levelled off, suggesting a carrying capacity of the fenced area of less than 300 individuals. Ottewell et al’s (2014) population viability analysis for the fenced population after 100 years estimates the population will decline from about 260 individuals to about 250 individuals (Figure 2), but genetic heterozygosity will decline linearly to about 80% diversity of the source population and allelic diversity will decline faster to about 50% after 100 years.
Figure 2. Population viability analysis of the fenced Golden Bandicoots at Lorna Glen (Ottewell presentation at Genetic Diversity workshop 12/5/16)
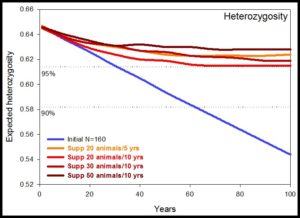
So if the population was augmented by new individuals would that increase heterozygosity and allelic richness after 100 years? The answer is yes, and 20 new animals per 10 years is required to maintain heterozygosity (Figure 3) and 50 new individuals per 10 years is required to maintain allelic richness (Figure 4).
Figure 3. Heterozygosity in Golden Bandicoots in the fenced compound of Lorna Glen with various augmentations (Ottewell presentation at Genetic Diversity workshop 12/5/16)
Figure 4. Allelic richness in Golden Bandicoots in the fenced compound of Lorna Glen with various augmentations (Ottewell presentation at Genetic Diversity workshop 12/5/16)
If Golden Bandicoots were released outside the fence and the population increased to 1,000 individuals, would this improve heterozygosity and allelic diversity? Yes, but there is still a slow progressive decline and the population would have to be augmented from an outside source, though smaller numbers of animals would be needed, and less frequently.
So what are the options for the DPaW in the ongoing management of this semi-wild population? If long-term population size remains low, replenishing genetic diversity from the Barrow Island population might assist, and modelling could help define an appropriate strategy. Other potential source populations are Marchinbar Island and the Dampier Peninsula in the Kimberley, which could introduce novel genetic diversity to the population at Lorna Glen but there could be a potential problem of outbreeding depression (Frankham et al. 2011). Introducing new genetic diversity into a declining population has proved successful in the case of the mountain pygmy possum, improving fitness and population growth. Continually augmenting the population with new individuals is costly, and this expenditure would need to be balanced with the potential use of funding for managing other threatened species. However, if strategies become available that allow an increase in population size to greater than 1000 individuals, genetic augmentation may not be needed.
So what is the future for the large number of translocated threatened species DPaW is planning to release on Dirk Hartog Island? DPaW is planning, as part of its Return to 1616 project to release 10 threatened species of mammals (i.e. the western barred bandicoot, chuditch, mulgara, dibbler, greater stick-nest rat, desert mouse, Shark Bay mouse, heath mouse, woylie and boodie) to Dirk Hartog Island once the feral and pest predators have been removed. Dirk Hartog Island is large (62,000 ha) so is likely to be able to sustain large populations of most of these species, however, in many cases the genetic diversity of the source populations for these species is much lower than the Golden Bandicoot on Barrow Island. Mixing multiple source populations as part of the release program to maximise the representation of genetic diversity in each of the species is seen as a way to reduce the need for genetic augmentation in the longer term. The management rationale behind these broadscale translocations to Dirk Hartog Island is to provide ‘safety populations’ of threatened species.
In addition to implementing significant biosecurity arrangements (Asher and Morris 2015) will there be an ongoing cost associated with species augmentation? Is there another way, and should these long-term costs be clearly articulated at the commencement of these relocation programs? Genetic analysis and population viability modelling can provide tools to help understand the long term trajectory of translocated populations and to evaluate appropriate strategies for their management.
We thank Kym Ottewell for assisting with the preparation of this post.
References
Asher, J., and K. Morris. 2015. Dirk Hartog Island Biosecurity Implementation Plan. Perth.
Frankham, R., J. D. Ballou, M. D. Eldridge, R. C. Lacy, K. Ralls, M. R. Dudash, and C. B. Fenster. 2011. Predicting the probability of outbreeding depression. Conservation Biology 25:465-475.
Ottewell, K., J. Dunlop, N. Thomas, K. Morris, D. Coates, and M. Byrne. 2014. Evaluating success of translocations in maintaining genetic diversity in a threatened mammal. Biological Conservation 171:209-219.
Photo credit: Golden Bandicoot (Supplied by Dr Kym Ottewell)
Comments
Got something to say?